Abstract
5-alpha reductase inhibitors (5-ARIs), including finasteride and dutasteride, are well established in the medical management of benign prostatic hyperplasia (BPH). Beyond their effects on prostate volume, 5-ARIs exert a profound antiangiogenic effect on prostatic tissue through multiple interconnected mechanisms. By inhibiting the conversion of testosterone to dihydrotestosterone (DHT), 5-ARIs suppress the expression of vascular endothelial growth factor (VEGF), reduce microvessel density (MVD), induce endothelial cell apoptosis, and modulate additional proangiogenic mediators including hypoxia-inducible factor-1α (HIF-1α). These effects translate into measurable clinical benefit, particularly in reducing perioperative bleeding during transurethral resection of the prostate (TURP), controlling BPH-related haematuria, and treating refractory hematospermia. The same antiangiogenic rationale may also be relevant to modern enucleation procedures. Importantly, the vascular regression induced by 5-ARIs occurs rapidly — within as few as two weeks — preceding volumetric gland reduction. This review synthesises the current evidence on the molecular mechanisms underlying 5-ARI-induced prostatic devascularisation, with emphasis on its translational relevance to surgical and clinical urological practice.
1. Introduction
Benign prostatic hyperplasia (BPH) is one of the most prevalent urological conditions affecting aging men, with histological evidence present in up to 80–90% of men in their seventh and eighth decades of life [1]. The 5-alpha reductase inhibitors (5-ARIs) finasteride and dutasteride represent a cornerstone of medical therapy for BPH, acting by blocking the intraprostatic conversion of testosterone to dihydrotestosterone (DHT), the principal androgen responsible for prostate growth and development [2][3].
While 5-ARIs are primarily recognised for their ability to reduce prostate volume by 20–30% and decrease the risk of acute urinary retention and BPH-related surgery [4][5], an increasingly well-characterised but underappreciated effect is their capacity to reduce prostatic vascularity. This antiangiogenic property has direct clinical relevance: a recent meta-analysis of 30 randomised controlled trials by Hehir et al. (2026) demonstrated that preoperative 5-ARI administration significantly reduces intraoperative blood loss (mean difference −82.58 mL), haemoglobin drop (−0.90 g/dL), blood transfusion rates (OR 0.31), and operative time during TURP [6].
The purpose of this review is to delineate the molecular and cellular mechanisms through which 5-ARIs achieve prostatic vascular regression, and to discuss the translational implications for surgical practice — including their potential relevance to modern enucleation techniques.
2. The DHT–VEGF Axis: The Central Mechanism
The primary mechanism through which 5-ARIs reduce prostatic vascularity is the disruption of the androgen-driven VEGF signalling pathway. Under physiological and hyperplastic conditions, DHT acts as a potent stimulator of VEGF expression in prostatic stromal and epithelial cells [7][8]. VEGF, in turn, is the dominant proangiogenic factor responsible for driving neovascularisation within the hyperplastic prostate [9].
The intraprostatic concentration of DHT is approximately five-fold higher than that of testosterone, owing to the activity of 5-alpha reductase [10][11]. Finasteride, which selectively inhibits the type 2 isoenzyme, reduces serum DHT by approximately 70%, while dutasteride, a dual inhibitor of types 1 and 2, achieves suppression exceeding 90–95% [12][13]. This reduction in intraprostatic DHT leads to a significant downregulation of VEGF expression in prostatic tissue.
Pareek et al. (2003) demonstrated that finasteride-treated patients undergoing TURP exhibited significantly lower VEGF expression and microvessel density (MVD) in suburethral prostatic tissue compared to untreated controls (p < 0.05) [14]. Importantly, the reduction in VEGF was most pronounced in the suburethral compartment, precisely the tissue most relevant to perioperative bleeding during transurethral procedures.
Häggström et al. (1999) had previously shown in a castration model that testosterone directly induces VEGF synthesis in the ventral rat prostate, establishing the mechanistic link between androgen signalling and prostatic angiogenesis [15]. The 5-ARIs replicate this effect pharmacologically by reducing DHT without complete androgen deprivation.
3. Microvessel Density Reduction: Evidence from Human Studies
Multiple clinical studies have confirmed that 5-ARI treatment reduces MVD in human prostatic tissue. Hochberg et al. (2002) first reported significantly decreased suburethral MVD in finasteride-treated prostates using CD34 immunohistochemistry [16]. This finding was subsequently confirmed by Memis et al. (2008), who demonstrated reduced MVD specifically in the suburethral zone after 4 weeks of finasteride therapy [17].
A critical finding from Donohue et al. (2005) was that even a short 2-week course of finasteride can significantly reduce prostatic MVD and VEGF expression in a randomised, placebo-controlled setting. In 64 patients randomised to finasteride 5 mg or placebo before TURP, MVD was 60 vs 71 and VEGF scores were 47 vs 61 (p < 0.01 and p < 0.001, respectively) [18b]. This rapid effect — which precedes meaningful volumetric gland reduction — indicates that the antiangiogenic mechanism of 5-ARIs is independent of their gland-shrinking properties and represents a primary pharmacological effect.
The meta-analysis by Hehir et al. pooled data from multiple RCTs and confirmed a significant reduction in MVD (MD = −6.18 vessels/mm³, p < 0.001) and VEGF expression (MD = −3.25, p < 0.001) in 5-ARI-treated specimens, providing level 1 evidence for this effect [6].
4. Endothelial Cell Apoptosis and Vascular Regression
Beyond the suppression of proangiogenic signalling, 5-ARIs actively promote regression of existing prostatic microvasculature through induction of apoptosis. Sutton et al. (2006) demonstrated that finasteride treatment leads to a significant increase in the apoptotic index (by TUNEL assay) and reduced MVD (Factor VIII staining) in prostatic specimens from BPH patients treated for 1–12 months compared to untreated controls (p < 0.01) [19].
The same study showed that finasteride inhibits prostate epithelial cell adhesion in vitro, suggesting an additional mechanism by which the drug disrupts the structural integrity of prostatic tissue and its vascular support network. The authors proposed that finasteride targets prostate vascularity through a dual mechanism: inducing apoptosis in both endothelial and epithelial compartments, and inhibiting cell–cell adhesion interactions necessary for vascular maintenance [19].
Rittmaster et al. (1995) provided earlier evidence of finasteride-induced apoptosis and atrophy in the ventral rat prostate, demonstrating that 5-ARI treatment leads to ductal atrophy through programmed cell death rather than simple quiescence [20]. This apoptotic process in the epithelial and stromal compartments reduces the metabolic demand of prostatic tissue, further diminishing the paracrine signalling that sustains the microvascular network.
5. Modulation of HIF-1α and Additional Angiogenic Mediators
Lekas et al. (2006) evaluated the effects of finasteride on hypoxia-inducible factor-1α (HIF-1α), VEGF, and MVD in resected prostatic tissue from BPH patients. They found statistically significant reductions in all three parameters in finasteride-treated specimens compared to controls [21]. HIF-1α is a master transcription factor that responds to tissue hypoxia by upregulating a suite of proangiogenic genes, including VEGF, fibroblast growth factor (FGF), and platelet-derived growth factor (PDGF) [22].
The reduction of HIF-1α by finasteride suggests that 5-ARIs may interrupt a broader hypoxia-driven angiogenic programme in the hyperplastic prostate, rather than acting solely through VEGF suppression. In hyperplastic prostatic tissue, the expanding adenoma creates areas of relative hypoxia that stimulate HIF-1α-mediated neovascularisation. By simultaneously reducing both the tissue mass (and therefore hypoxic demand) and the direct androgen-driven VEGF pathway, 5-ARIs achieve a synergistic antiangiogenic effect.
Ku et al. (2009) extended these findings to dutasteride, demonstrating similar reductions in HIF-1α and VEGF expression in both rat and human prostatic tissue, supporting a class effect rather than a finasteride-specific phenomenon [23].
Table 1
| Study / Year |
Design |
Treatment |
Duration |
N |
Key findings |
Compartment |
Key p-value |
Ref |
| Pareek et al. 2003 |
Observational comparative |
Finasteride 5 mg |
Variable (pre-TURP) |
~24 |
↓ VEGF expression and ↓ suburethral MVD (CD34) |
Suburethral |
p < 0.05 |
[14] |
| Hochberg et al. 2002 |
Comparative |
Finasteride |
Variable |
~20–30 |
↓ Suburethral MVD (CD34) in haematuria/BPH patients |
Suburethral |
Significant |
[16] |
| Memis et al. 2008 |
Prospective comparative |
Finasteride 5 mg |
4 weeks |
30 |
↓ Significant suburethral MVD vs controls |
Suburethral |
Significant |
[17] |
| Donohue et al. 2005 |
Placebo-controlled RCT |
Finasteride 5 mg |
2 weeks |
64 |
↓ MVD (60 vs 71) and ↓ VEGF (47 vs 61) in post-TURP tissue |
Prostatic (post-TURP) |
MVD p < 0.01; VEGF p < 0.001 |
[18b] |
| Lekas et al. 2006 |
Comparative |
Finasteride |
Variable |
Not spec. |
↓ HIF-1α, ↓ VEGF, ↓ MVD in resected tissue |
Prostatic |
Significant |
[21] |
| Sutton et al. 2006 |
Comparative + in vitro |
Finasteride |
1–12 months |
27 |
↑ Apoptotic index (TUNEL), ↓ MVD (Factor VIII), ↓ cell adhesion |
Prostatic |
p < 0.01 |
[19] |
| Ku et al. 2009 |
Comparative (rat + human) |
Dutasteride |
Variable |
N/A (mixed model) |
↓ HIF-1α and ↓ VEGF in human and rat prostatic tissue |
Prostatic |
Significant |
[23] |
| Hehir et al. 2026 |
Meta-analysis (30 RCTs) |
5-ARIs (fin/dut) |
Variable (incl. 2 wk) |
2974 |
↓ MVD (MD −6.18 vessels/mm³); ↓ VEGF (MD −3.25) |
Prostatic (pooled) |
p < 0.001 both |
[6] |
Table 1. Summary of key evidence on 5-ARI-induced reduction of microvessel density (MVD) and VEGF expression in human prostatic tissue. MD = mean difference.
[18b] Donohue JF, et al. Randomized, placebo controlled trial showing that finasteride reduces prostatic vascularity rapidly within 2 weeks. BJU Int. 2005;96(9):1319–1322.
6. A Bidirectional Model: Tissue Involution and Vascular Regression
The relationship between prostatic tissue involution and vascular regression is bidirectional. On one hand, reduction of DHT leads to epithelial and stromal apoptosis, which decreases the metabolic demand and paracrine signalling that sustain the microvascular bed [20][24]. On the other hand, microvascular regression reduces the trophic support available to the glandular tissue, further accelerating involution [19].
This positive feedback loop explains why the antiangiogenic effects of 5-ARIs are detectable before significant volumetric reduction occurs: endothelial cell apoptosis is a relatively rapid process (days to weeks), whereas glandular involution requires sustained androgen suppression over months [18b][25]. The clinical implication is that even short preoperative courses of 5-ARIs (as little as 2 weeks) may confer haemostatic benefit during surgery, as confirmed by the meta-analytic data [6].
It is worth noting that 5-alpha reductase inhibition reduces prostatic size by 20–30% through induction of apoptosis, histologically manifested as ductal atrophy, and diminishes the number of blood vessels through VEGF reduction [25]. This dual action — volumetric and vascular — distinguishes 5-ARIs from pure alpha-blockers, which provide symptomatic relief without altering the underlying tissue biology.
7. Application in Hematospermia: Extension of the Antiangiogenic Rationale
An additional clinical application that directly reflects the antiangiogenic properties of 5-ARIs is the treatment of hematospermia (hemospermia). Although hematospermia is most commonly a benign, self-limiting condition, persistent or recurrent cases can cause significant patient distress and pose a diagnostic challenge [27]. The seminal vesicles and prostatic urethra share the same androgen-dependent vascular microenvironment as the transition zone; consequently, the mechanisms of MVD reduction and VEGF suppression described above are equally relevant to this clinical scenario.
Badawy et al. (2012) conducted a prospective, placebo-controlled study evaluating finasteride 5 mg daily for 3 months in 24 patients with idiopathic refractory hematospermia. In the finasteride group, 66.7% of patients experienced complete remission of bleeding episodes within 2–5 weeks, confirmed objectively by semen analysis. In contrast, only 25% of placebo-treated patients showed improvement, and those who did still demonstrated significant residual erythrocytes on microscopy (>50 RBC/HPF). No recurrence was observed during the 3-month treatment period in responders [28].
This rapid response — within weeks, not months — mirrors the timeline of MVD reduction observed in the preoperative TURP studies, reinforcing the concept that the antiangiogenic effect of 5-ARIs precedes gland involution and operates as the primary mechanism of bleeding control.
Zhang et al. (2014) reported on the combination of transurethral seminal vesiculoscopy (TUSV) with perioperative finasteride (5 mg/d for 2 weeks pre- and post-procedure) in 32 patients with recurrent hematospermia. The combined approach allowed both diagnostic evaluation and treatment, with finasteride providing a pharmacological reduction in tissue vascularity that complemented the endoscopic intervention [29].
In the management algorithms for hematospermia, 5-ARIs are now positioned as a pharmacological option for persistent cases after exclusion of infection and malignancy, particularly when the bleeding source is prostatic or seminal vesicular in origin [30][31]. The rationale is mechanistically identical to the perioperative use in TURP: by reducing DHT-driven VEGF expression and MVD in the prostatic and periprostatic vasculature, finasteride diminishes the fragility and density of submucosal vessels prone to rupture during erection and ejaculation.
A noteworthy paradox exists: finasteride has also been reported as a rare cause of hematospermia, particularly in younger men taking low-dose (1 mg) finasteride for androgenetic alopecia [32]. This apparent contradiction may reflect the transitional vascular remodelling that occurs during the initial phase of treatment, before a new steady-state of reduced vascularity is established. The clinical significance of this paradoxical effect appears to be minimal and self-limiting.
8. Clinical and Surgical Implications
Preoperative optimisation for TURP: The meta-analysis by Hehir et al. (2026) provides robust evidence that preoperative 5-ARI administration reduces intraoperative blood loss, transfusion requirements, irrigation volume, and operative time during TURP. Even short courses of 2 weeks appear to be effective, making this a practical preoperative intervention [6].
Management of BPH-related haematuria: Kearney et al. demonstrated that finasteride effectively controls gross haematuria secondary to BPH, with 94% of patients experiencing improvement and 77% achieving complete resolution, regardless of anticoagulation status [26]. This effect is mediated by the MVD reduction in suburethral prostatic tissue.
Potential relevance to enucleation procedures: While the current evidence is largely based on TURP, the antiangiogenic mechanisms of 5-ARIs should theoretically benefit any procedure that involves transurethral dissection of prostatic tissue. During holmium laser enucleation of the prostate (HoLEP) and other enucleation techniques, bleeding from the capsular plane is a significant intraoperative consideration. Whether preoperative 5-ARI therapy confers similar haemostatic advantages during enucleation as it does during resection remains an important area for future investigation.
Patients on anticoagulation: 5-ARIs may be particularly valuable in patients who require ongoing anticoagulant or antiplatelet therapy and are at increased risk of perioperative bleeding. The reduction in tissue vascularity may partially offset the systemic haemostatic impairment in these patients.
Figure 1. Integrated Model of 5-ARI-Induced Prostatic Vascular Regression
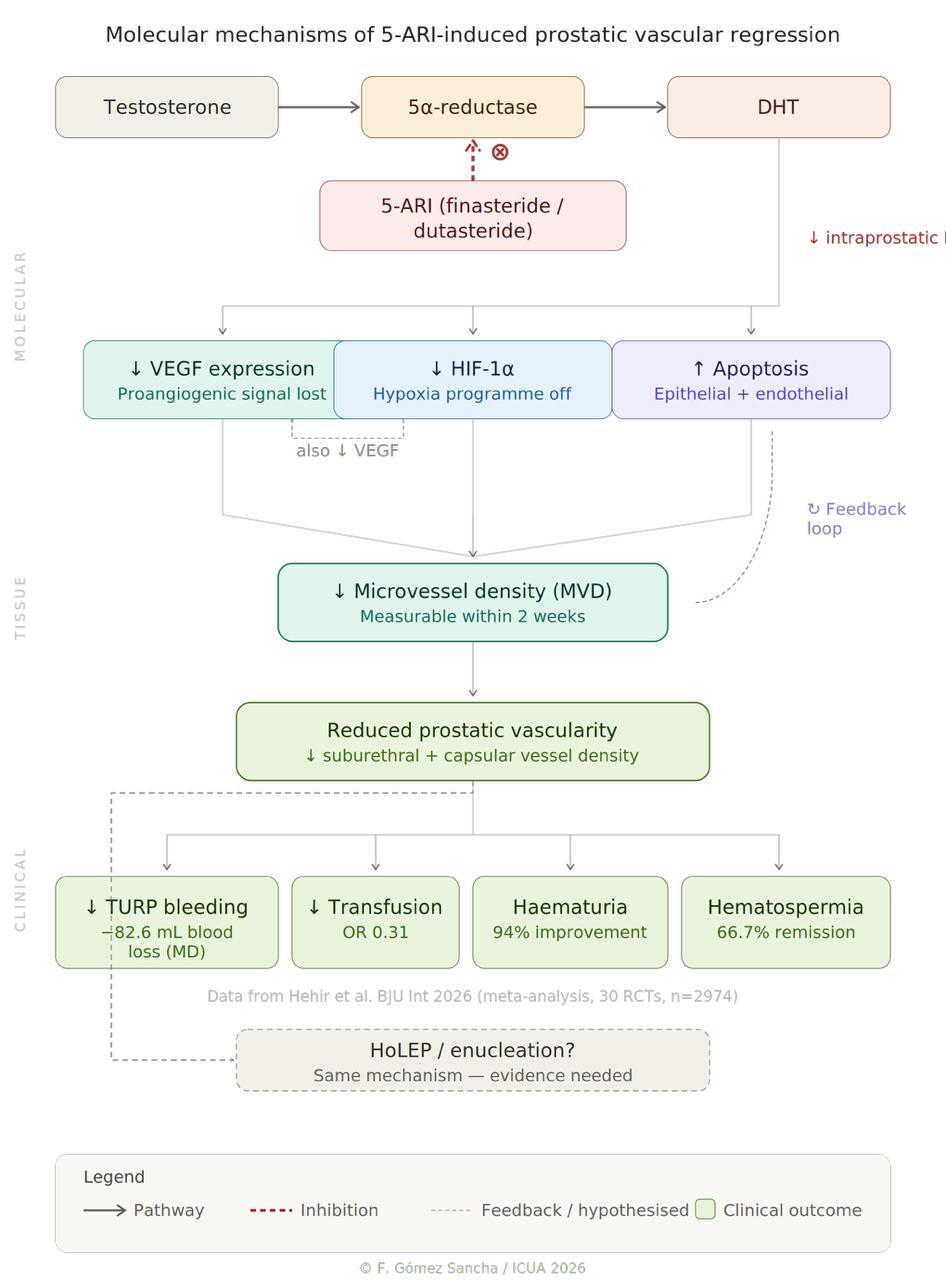
Figure 1. Three-level model of 5-ARI-induced prostatic devascularisation: molecular pathways, tissue-level effects, and clinical outcomes.
9. Conclusion
5-alpha reductase inhibitors reduce prostatic vascularity through a cascade of interconnected mechanisms: suppression of DHT-driven VEGF expression, reduction of microvessel density, induction of endothelial and epithelial apoptosis, and modulation of the broader HIF-1α-mediated angiogenic programme. These effects occur rapidly, with measurable MVD reduction within 2 weeks of treatment, and are supported by level 1 evidence from pooled randomised controlled trials.
Understanding these mechanisms is important not only for optimising perioperative management in BPH surgery but also for informing clinical decision-making regarding the timing and duration of preoperative 5-ARI therapy. Future research should evaluate whether these antiangiogenic benefits extend to modern enucleation techniques, and whether specific patient subgroups (e.g., those on anticoagulation, or with highly vascularised prostates) derive disproportionate benefit from preoperative 5-ARI administration.
References
[1] Roehrborn CG. Pathology of benign prostatic hyperplasia. Int J Impot Res. 2008;20 Suppl 3:S11–S18. PubMed
[2] Rittmaster RS. 5alpha-reductase inhibitors in benign prostatic hyperplasia and prostate cancer risk reduction. Best Pract Res Clin Endocrinol Metab. 2008;22(2):389–402. PubMed
[3] Traish AM, Mulgaonkar A, Giordano N. The dark side of 5α-reductase inhibitors' therapy. Korean J Urol. 2014;55(6):367–379. PubMed
[4] McConnell JD, et al. The long-term effect of doxazosin, finasteride, and combination therapy on the clinical progression of BPH. N Engl J Med. 2003;349(25):2387–2398. PubMed
[5] Roehrborn CG, et al. Efficacy and safety of dutasteride in men with BPH. Urology. 2002;60(3):434–441. PubMed
[6] Hehir CM, et al. The role of 5-alpha reductase inhibitors in TURP: a meta-analysis of RCTs. BJU Int. 2026. doi: 10.1111/bju.70117. PubMed
[7] Chislett B, et al. 5-ARI use in prostatic disease and beyond. Transl Androl Urol. 2023;12(3):487–505. PMC
[8] Jackson MW, et al. VEGF expression in prostate cancer and BPH. J Urol. 1997;157(6):2323–2328. PubMed
[9] Ferrara N. Role of VEGF in the regulation of angiogenesis. Kidney Int. 1999;56(3):794–814. PubMed
[10] Bruchovsky N, Wilson JD. The conversion of testosterone to 5α-DHT by rat prostate. J Biol Chem. 1968;243(8):2012–2021. PubMed
[11] Carson C, Rittmaster R. The role of DHT in BPH. Urology. 2003;61(4 Suppl 1):2–7. PubMed
[12] Clark RV, et al. Marked suppression of DHT by dutasteride. J Clin Endocrinol Metab. 2004;89(5):2179–2184. PubMed
[13] Bramson HN, et al. Unique preclinical characteristics of GG745, a potent dual inhibitor of 5AR. J Pharmacol Exp Ther. 1997;282(3):1496–1502. PubMed
[14] Pareek G, et al. Effect of finasteride on VEGF expression and microvessel density. J Urol. 2003;169(1):20–23. PubMed
[15] Häggström S, et al. Testosterone induces VEGF synthesis in the ventral prostate in castrated rats. J Urol. 1999;161(5):1620–1625. PubMed
[16] Hochberg DA, et al. Decreased suburethral prostatic MVD in finasteride treated prostates. J Urol. 2002;167(4):1731–1733. PubMed
[17] Memis A, et al. Effect of finasteride on suburethral prostatic MVD in hematuria patients. Urol Int. 2008;80(2):177–180. PubMed
[18] Donohue JF, et al. TURP and bleeding: RCT of finasteride for decreasing blood loss. J Urol. 2002;168(5):2024–2026. PubMed
[18b] Donohue JF, et al. Finasteride reduces prostatic vascularity rapidly within 2 weeks. BJU Int. 2005;96(9):1319–1322. PubMed
[19] Sutton M, et al. Finasteride targets prostate vascularity by inducing apoptosis and inhibiting cell adhesion. Prostate. 2006;66(11):1194–1202. PubMed
[20] Rittmaster RS, et al. Evidence for atrophy and apoptosis in the rat prostate with finasteride. Endocrinology. 1995;136(2):741–748. PubMed
[21] Lekas AG, et al. Finasteride effects on hypoxia and angiogenetic markers in BPH. Urology. 2006;68(2):436–441. PubMed
[22] Semenza GL. Targeting HIF-1 for cancer therapy. Nat Rev Cancer. 2003;3(10):721–732. PubMed
[23] Ku JH, et al. Effect of dutasteride on HIF-1α, VEGF and MVD in rat and human prostate tissue. Scand J Urol Nephrol. 2009;43(6):445–453. PubMed
[24] Tian HL, et al. Finasteride reduces MVD and VEGF in renal tissue of diabetic rats. Am J Med Sci. 2015;349(6):516–520. PubMed
[25] Naslund MJ, Miner M. Clinical efficacy and safety of 5-ARIs for the enlarged prostate. Clin Ther. 2007;29(1):17–31. PubMed
[26] Kearney MC, et al. Finasteride for control of gross hematuria due to BPH. J Urol. 2002;167(6):2489–2491. PubMed
[27] Ahmad I, Krishna NS. Hemospermia. J Urol. 2007;177(5):1613–1618. PubMed
[28] Badawy AA, et al. Finasteride for treatment of refractory hemospermia: prospective placebo-controlled study. Int Urol Nephrol. 2012;44(2):371–375. PubMed
[29] Zhang K, et al. Transurethral seminal vesiculoscopy combined with finasteride for recurrent hematospermia. Zhonghua Nan Ke Xue. 2014;20(5):449–452. PubMed
[30] Efesoy O, et al. Novel algorithm for the management of hematospermia. Urol Res Pract. 2023;49(6):398–405.
[31] Mathers MJ, et al. Hematospermia: etiology, diagnosis, and treatment. Dtsch Arztebl Int. 2017;114(45):757–762. PubMed
[32] Alharbi FF, et al. Hematuria and hematospermia associated with finasteride for androgenic alopecia. Drug Saf Case Rep. 2017;4(1):14. PubMed